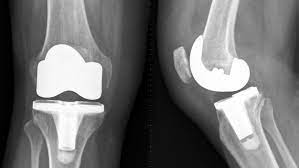
In February 2022, Exactech recalled tens of thousands of lower-leg and ankle joint replacing items which had been loaded into folks between 2004 and 2022. The joint and foot inserts skilled second-rate packing that did not shield the numerous factors from contact with air flow before simply being implanted into the overall body. Oxidation might cause the inserts to significantly degrade as time passes, departing quite a few people in ache and suffering from the potential risk of revision surgical treatment.
WHY WAS THE EXACTECH KNEE IMPLANT RECALLED?
The remember for Defective Exactech Knee Replacement alternatives is due to a wrapping breaking down. It really is essential for that inserts not to arrive in contact with air prior to getting placed in the program (as an example, even though becoming provided and kept before your knee medical procedure). The packaging levels for this plastic-kind inserts in this remember have been discovered to get allowed air movement before placement, which causes oxidation and untimely dress yourself in. The constituents and inserts built over the past 8 yrs were actually actually manufactured in o2 tolerant vacuum luggage but happen to be absent a barrier addressing that contain ethylene vinyl liquor (EVOH) that further more much more augments o2 opposition.
Plenty of Exactech affected individuals have received a really concerning discover through the medical professional and also have a large amount of issues. A system remember leads to patients considering whether or not they have an Exactech knee or foot alternative and, if they do, what activities they should decide to use decide if the remember consequences them separately. At Searcy Denney, our experienced flawed health-related product authorized experts might help locate these responses yourself and determine if fiscal settlement could be designed for yourself and your family as a consequence of an Exactech lower body replacement court action.